|
Rotation and rotation-vibration spectra in rare-gas matrices Mason, von Holle, et al., 1971. Laser emission in the pure rotation spectrum Deutsch, 1967. Molecular constants Bondybey, Pearson, et al., 1972, Krauss and Neumann, 1974, Meyer and Rosmus, 1975, Dunning, 1976. RKR potential curves Fallon, Vanderslice, et al., 1960, di Londardo and Douglas, 1973, Dunham potential coefficients Webb and Rao, 1968, Ogilvie and Koo, 1976. 
The dissociation energy 47333 ± 60 cm -1 has been determined di Londardo and Douglas, 1973. A few broadened lines near these limits have been observed di Londardo and Douglas, 1973. All levels up to the last (v=19) are tabulatedįor v=14.19 the rotational levels break off at decreasing J on account of predissociation by Higher vibrational levels (v≤19) was derived by Johns and Barrow, 1959. 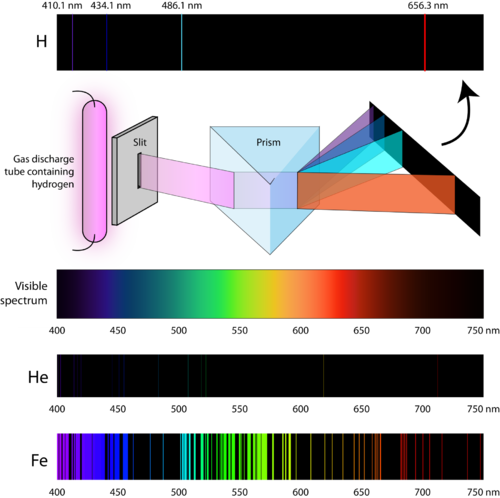
Introduction of the Dunham correction Webb and Rao, 1968 gives ω e = 4138.767 Webb and Rao, 1968 and B e = ( 3Π, 1Π, 3Σ +) arising from ground state atomic products were given Theoretical potential curves for three repulsive states HF is quite transparent to 1650 Safari, 1954. The B (or V) state was also observed in the electronĮnergy loss spectrum Salama and Hasted, 1976. Strong perturbations above v'=27, butīands have been identified to v'=73. Very extensive band system (also called V-X) extending in absorption from 96000 toġ17000, in emission from 36000 to 70000 cm -1. RKR potential curves Fallon, Vanderslice, et al., 1960, di Londardo and Douglas, 1973. Vibrational levels di Londardo and Douglas, 1973. The vibrational and rotational constants, were obtained from a fit to the seven lowest Two strong bands between 104000 cm -1, not yet analysed.Ībsorption bands above 100000 cm -1, not yet analysed. ↳ Weiss, 1963 Muenter and Klemperer, 1970 Muenter, 1972 de Leeuw and Dymanus, 1973 ↳ Rothschild, 1964 Revich and Stankevich, 1966 Mason and Nielsen, 1967 ↳ Mann, Thrush, et al., 1961 Herget, Deeds, et al., 1962 Fishburne and Rao, 1966 Webb and Rao, 1968 ↳ Johns and Barrow, 1959 missing citationĬontinuous absorption starting at 60600 cm -1. ↳ di Lonardo and Douglas, 1972 di Londardo and Douglas, 1973 Rydberg levels converging to the ground state of HF + have been observed Position of 0-0 band (units noted in table) Observed transition(s) corresponding to electronic state Rotational constant – first term, centrifugal force (cm -1) Rotation-vibration interaction constant (cm -1) Rotational constant in equilibrium position (cm -1) Vibrational constant – third term (cm -1) Vibrational constant – second term (cm -1) Vibrational constant – first term (cm -1) Symbols used in the table of constants SymbolĮlectronic state and / or symmetry symbol 
Secretary of Commerce on behalf of the U.S.A.ĭata compiled by: Klaus P. Your institution may already be a subscriber.įollow the links above to find out more about the dataīy the U.S. With the development of data collections included in The purpose of the fee is to recover costs associated NIST subscription sites provide data under theĭata Program, but require an annual fee to access. NIST / TRC Web Thermo Tables, professional edition (thermophysical and thermochemical data).Computational Chemistry Comparison and Benchmark Database.Microwave spectra (on physics lab web site).Use this link for bookmarking this species This structure is also available as a 2d Mol file IUPAC Standard InChIKey: KRHYYFGTRYWZRS-UHFFFAOYSA-N Copy.The Rydberg formula remained unexplained until the first successful model of the hydrogen atom was proposed in 1913. The Rydberg formula for hydrogen gives the exact positions of the spectral lines as they are observed in a laboratory however, at the beginning of the twentieth century, nobody could explain why it worked so well. As you may guess, there are infinitely many such spectral bands in the spectrum of hydrogen because \(n_f\) can be any positive integer number. When \(n_f = 6\), we have the Humphreys series. When \(n_f = 5\), the series is called the Pfund series. When \(n_f = 4\), the series is called the Brackett series. When \(n_f = 3\), the series is called the Paschen series.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
